Recently, the research team led by Prof. Wang Lianhui and Prof. Wang Ting from the State Key Laboratory of Flexible Electronics and the School of Chemistry and Life Sciences at NJUPT has made significant progress in the field of flexible electrochemical sensing. They developed a novel flexible bioelectronic sensing system for in-situ, real-time, high-fidelity, and multi-modal monitoring of wounds. On October 7, their research article titled “Soft bioelectronics embedded with self-confined tetrahedral DNA circuit for high-fidelity chronic wound monitoring” was published in the international academic journal Nature Communications. Postdoctoral researcher Zhao Xiao is the first author of the paper, with Prof. Wang Lianhui and Prof. Wang Ting serving as the co-corresponding authors.

The assessment and treatment of chronic wounds is a major challenge in clinical medicine. Proteins at the wound site, especially inflammation-related proteins, are key indicators of healing progress. However, their extremely low concentration in wound exudate, combined with the complex wound environment, has led to common limitations in existing biosensors: insufficient sensitivity, vulnerability to human activity interference, performance degradation during long-term use, and easy contamination. To address these challenges, the team developed a flexible biosensor embedded with a Self-Confined Tetrahedral DNA Circuit (SCTD), which enables comprehensive, high-fidelity monitoring of stage-specific proteins and biophysical indicators in wounds (Fig. 1a). By using patterned wettable nanofiber patches, the system concentrates wound exudate and prevents the diffusion of auxiliary DNA, forming a hydrophilic biosensing reaction pool without impeding tissue regeneration (Fig. 1b). Through strategies of pre-fixing exogenous auxiliary nucleic acids and spatial confinement, the SCTD achieves target protein-triggered signal amplification (Fig. 1c), reducing the detection limit by one order of magnitude. The tetrahedral DNA structure ensures high-fidelity detection: signals vary within 3% after 1,000 bending cycles and show less than 8% attenuation over four weeks. Additionally, its anti-biological contamination capability is improved by over 50%, effectively resisting interference from the complex wound environment.
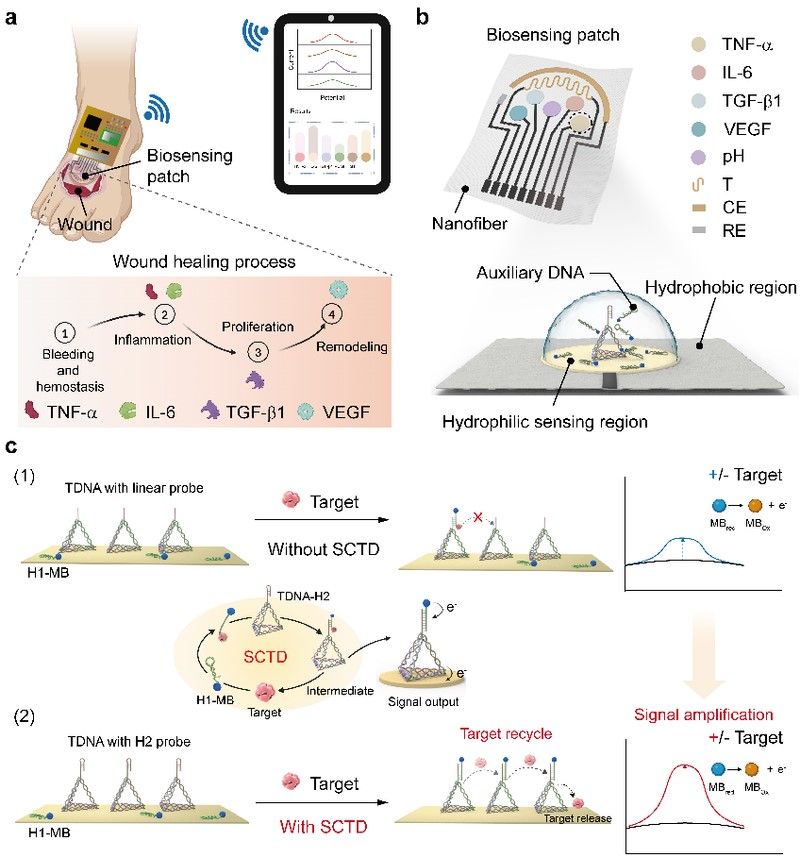 Schematic Diagram of Wearable Flexible Biosensing System Based on Nucleic Acid Cascaded Amplification for In-Situ Chronic Wound Monitoring
Schematic Diagram of Wearable Flexible Biosensing System Based on Nucleic Acid Cascaded Amplification for In-Situ Chronic Wound Monitoring
Integrated with a wireless communication module, this platform can simultaneously monitor multiple wound healing-related proteins (TNF-α, IL-6, TGF-β1, VEGF) and key physiological parameters (pH value, temperature) (Figs. 2a–c). The wireless sensing platform enables accurate, in-situ monitoring of both non-infected and infected wounds in diabetic mice without interfering with healing process (Figs. 2d, e). Compared with traditional wound monitoring methods, this flexible sensing system improves sensitivity and stability while enabling real-time, multi-parameter detection. In the future, it is expected to guide treatment decisions based on molecular characteristics and integrate with smart dressings to achieve responsive therapy.
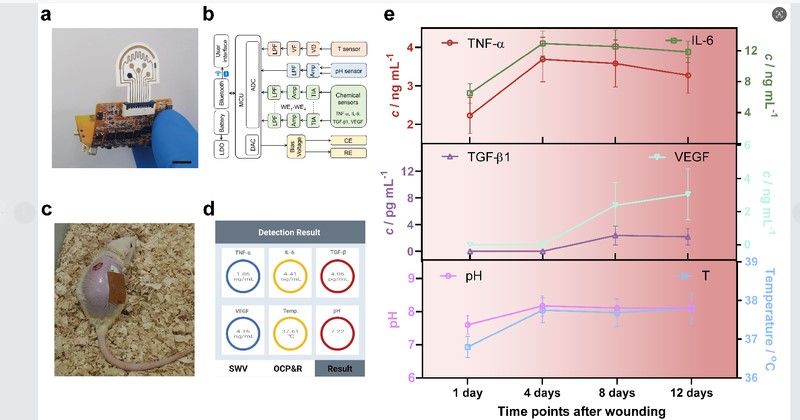
In-Situ Monitoring of Chronic Wounds by Wearable Sensing System in Diabetic Mouse Models
This research was supported by several programs, including the Excellent Young Scientist Fund and the Young Scientist Fund of the National Natural Science Foundation of China, the Jiangsu Provincial Outstanding Postdoctoral Program, and the High-Level Faculty Research Startup Program of Nanjing University of Posts and Telecommunications.
(Author: Zhao Xiao; Initial Review: Qiao Zuqin, Dai Xiucheng; Editor: Wang Cunhong; Final Review: Zhang Feng)



